Categories
- Global News Feed
- Uncategorized
- Alabama Stem Cells
- Alaska Stem Cells
- Arkansas Stem Cells
- Arizona Stem Cells
- California Stem Cells
- Colorado Stem Cells
- Connecticut Stem Cells
- Delaware Stem Cells
- Florida Stem Cells
- Georgia Stem Cells
- Hawaii Stem Cells
- Idaho Stem Cells
- Illinois Stem Cells
- Indiana Stem Cells
- Iowa Stem Cells
- Kansas Stem Cells
- Kentucky Stem Cells
- Louisiana Stem Cells
- Maine Stem Cells
- Maryland Stem Cells
- Massachusetts Stem Cells
- Michigan Stem Cells
- Minnesota Stem Cells
- Mississippi Stem Cells
- Missouri Stem Cells
- Montana Stem Cells
- Nebraska Stem Cells
- New Hampshire Stem Cells
- New Jersey Stem Cells
- New Mexico Stem Cells
- New York Stem Cells
- Nevada Stem Cells
- North Carolina Stem Cells
- North Dakota Stem Cells
- Oklahoma Stem Cells
- Ohio Stem Cells
- Oregon Stem Cells
- Pennsylvania Stem Cells
- Rhode Island Stem Cells
- South Carolina Stem Cells
- South Dakota Stem Cells
- Tennessee Stem Cells
- Texas Stem Cells
- Utah Stem Cells
- Vermont Stem Cells
- Virginia Stem Cells
- Washington Stem Cells
- West Virginia Stem Cells
- Wisconsin Stem Cells
- Wyoming Stem Cells
- Biotechnology
- Cell Medicine
- Cell Therapy
- Diabetes
- Epigenetics
- Gene therapy
- Genetics
- Genetic Engineering
- Genetic medicine
- HCG Diet
- Hormone Replacement Therapy
- Human Genetics
- Integrative Medicine
- Molecular Genetics
- Molecular Medicine
- Nano medicine
- Preventative Medicine
- Regenerative Medicine
- Stem Cells
- Stell Cell Genetics
- Stem Cell Research
- Stem Cell Treatments
- Stem Cell Therapy
- Stem Cell Videos
- Testosterone Replacement Therapy
- Testosterone Shots
- Transhumanism
- Transhumanist
Archives
Recommended Sites
Monthly Archives: March 2017
Patient Voices: Type 2 Diabetes – New York Times
Posted: March 14, 2017 at 2:46 pm
New York Times | Patient Voices: Type 2 Diabetes New York Times Nearly 400 million people around the world have Type 2 diabetes, including about 28 million in the United States. Of those, as many as eight million don't know they have it. Type 2 diabetes can wreak havoc on the entire body, affecting everything from ... |
See the original post here:
Patient Voices: Type 2 Diabetes - New York Times
Posted in Diabetes
Comments Off on Patient Voices: Type 2 Diabetes – New York Times
JDRF works to raise Type 1 diabetes awareness – Clearfield Progress
Posted: March 14, 2017 at 2:46 pm
Some people believe they are healthy as horses, work out like fiends and run marathons or lift 500 pounds. However, here is a sobering message:
Those people are at the same risk of getting Type 1 diabetes as the not-so-fit folks who consider a short walk quality exercise. Type 1 diabetes doesnt care who it attacks. It can be a model, movie star, athlete or couch potato.
The Illinois Chapter of the Juvenile Diabetes Research Foundation is spending most of spring and summer preparing for huge fall and winter events, and during this relative down time, the group is trying to get the word out to as many people as possible that Type 1 diabetes is not just affecting juveniles any more. According to Illinois JDRF Director of Corporate and Marketing Partnerships Dana Snodgrass, 1.25 million people have Type 1, and its not just kids.
Unfortunately, its a growing disease and there is nothing you can do to prevent it, she says. Its not like its caused by you eating unhealthy foods or you are not exercising or you are overweight. You are born with it and you can be diagnosed with it at any age.
Now people are being diagnosed when they are 30, 40 and 50 years old. In the past, if you were an adult with Diabetes, you were diagnosed as Type 2. But now, more people are finding out its actually Type 1. You might be 50 years old, but for some reason, your body just stopped producing insulin. You had it all along.
Snodgrass says if its not diagnosed, the worst-case scenario could be a diabetic coma that can result in death.
If you see someone, you might not be able to tell they even have it, she says. Its something you have the rest of your life and its difficult to maintain. I dont think enough people know about it.
Another project for JDRF is the yearlong Bag of Hope program in which kids who are diagnosed with Type 1 learn about how to live with the disease. A teddy bear with Diabetes, Rufus, helps to get children through this ordeal.
There are books, measuring cups and a lot of other resources for children with Type 1, Snodgrass says. In Illinois, we partner with hospitals and physicians offices, and children and parents receive these Bags of Hope. It helps them after their first couple of days.
Once you are diagnosed, your entire life changes. You have to learn what you can eat and when you can eat it, how to give yourself injections, monitoring your blood. You are usually in the hospital for three days and you have all the resources there. Then you go home and all of this becomes the new normal. Its earthshattering. This bag is for the kids and parents to walk them through what needs to be done now that they are out of the hospital.
Later in the year, JDRF will host walkson September 24 and October 1that will celebratethe event's38th year and, in the past, have attracted 30,000-32,000 people per year.
Originally posted here:
JDRF works to raise Type 1 diabetes awareness - Clearfield Progress
Posted in Diabetes
Comments Off on JDRF works to raise Type 1 diabetes awareness – Clearfield Progress
Program offers help managing diabetes – Gaston Gazette
Posted: March 14, 2017 at 2:46 pm
From staff reports
Do you or a family member have diabetes and on Medicare? If so, then learn to take control of your diabetes with the upcoming "Living Healthy with Diabetes" workshop.
NC Cooperative Extension is offering the next session of theworkshop, a free self-management program which begins in May. This particular workshop is especially designed for Medicare Part B recipients (ages 65 and older) who have been diagnosed with diabetes. These individuals will be eligible for one-on-one nutrition counseling with a registered dietitian.
Were excited to be able to offer more personalized services with this session of Living Healthy with Diabetes , said Linda Minges, program facilitator for Gaston Extension. "Individuals who qualify for nutrition counseling with the dietitian will learn from other individuals with diabetes, as well as get personalized help with meal planning."
"Living Healthy with Diabetes" is a Stanford University program designed for anyone with diabetes, pre-diabetes, or at risk for diabetes. Participants will learn how to prevent low blood sugar; prevent and delay complications of diabetes; eat well; use medications effectively; manage pain, fatigue and depression; solve problems and set goals. The program is valued at more than $400 and there is no cost to participants.
'Living Healthy with Diabetes' truly makes a difference in the lives of so many people," said Minges. "Many participants report how helpful the group discussions are as well as being able to problem-solve together on health issues that impact everyone.
The program consists of six weekly sessions, scheduled for Wednesdays, May 10 through June 14 from 9 to11:30 a.m. All sessions will be held at the Lucile Tatum Center,959 Osceola St., Gastonia. There is no cost, but pre-registration is required. Participantsmust be able to attend at least four out of the six sessions. Participants receive a copy of Living a Healthy Life with Chronic Conditions workbook, a relaxation CD, tote bag, and refreshments. Pre-registration is required by contacting Linda J. Minges at 704-922-2127 or linda_minges@ncsu.edu before April 19. This program is supported by Centralina Area Agency on Aging.
Read the original:
Program offers help managing diabetes - Gaston Gazette
Posted in Diabetes
Comments Off on Program offers help managing diabetes – Gaston Gazette
3-D visualization of the pancreasnew tool in diabetes research – Medical Xpress
Posted: March 14, 2017 at 2:46 pm
March 14, 2017 The three-dimensional visualization, created with OPT, shows the pancreas of a healthy mouse. The individual pancreatic islets have been color-coded and their exact volume and 3-D-coordinates can be precisely determined throughout the pancreas. The exocrine pancreatic tissue (in grey) has partly been digitally removed. Credit: Ulf Ahlgren.
Ume researchers have created datasets that map the three-dimensional distribution and volume of the insulin-producing cells in the pancreas. The wealth of visual and quantitative information may serve as powerful reference resource for diabetes researchers. The Ume University researchers are now publishing their datasets in Scientific Data.
The hormone insulinwhich is needed to regulate the blood sugar levels of the bodyis produced by the pancreas and plays a key role in the development of diabetes. Insulin-producing cells are organised in the so-called Islets of Langerhans (or pancreatic islets), which are scattered by the thousands in the pancreas. In diabetes research, it is often important to study the quantity and distribution of insulin-producing cells. At present, such studies are generally based upon analyses of chosen cross-sections of pancreatic tissue. These in turn form the basis for attempting to gain an overall picture of the pancreas.
"However, such analyses only provide limited information and are often ridden with relatively large margins of error since the conclusions are based only on two-dimensional data," says Ulf Ahlgren, professor in molecular medicine at Ume University and in charge of the publications.
Ulf Ahlgren and his research colleagues at the Ume Centre for Molecular Medicine (UCMM) have previously developed new methods to create three-dimensional images of the insulin cell distribution in intact pancreas based on so-called optical projection tomography (OPT). This technique in many ways bears resemblance to a medical CT scanner, but instead of x-rays it uses regular light.
"We believe that the current publication represents the most comprehensive anatomical and quantitative description of the insulin cell distribution in the pancreas. By making these datasets accessible to other researchers, the data will be available for use as a powerful tool for a great number of diabetes studies. Examples may include planning of stereological analyses, in the development of non-invasive imaging techniques or various types of computational modelling and statistical analyses," says Ulf Ahlgren.
The video will load shortly
The datasets now published in Scientific Data consist of tomographic and 3D images. The datasets also include information on the individual volume of the Islets of Langerhans and their 3D coordinates and appearance throughout the entire pancreas in both healthy mice and obese mice (ob/ob), at different ages. The obese mice used in the study have a mutation that make them prone to develop obesity and diabetes.
The datasets highlight that islets differ in size and quantity within, and between, the various lobes of the pancreas. According to the research team, this emphasises that the pancreas should not be seen as a homogenous organ when experimental diabetes researchers study the insulin-producing Islets of Langerhans.
Visualising changes in the Islets of Langerhans
The datasets presented in Scientific Data form the basis of another recently published study in Scientific Reports. In that study, the researchers used the 3D data to identify changes in the Islets of Langerhans in the obese (ob/ob) mice. This animal model is often used to study initial metabolic changes that can lead to the development of type 2 diabetes. With the help of their refined techniques, the researchers could show that these mice to a great extent develop lesions in the Islets of Langerhans, manifesting as cyst-like structures. The study shows that these lesions are caused by internal bleeding as a consequence of an increased blood flow and instability of the blood vessels.
"Obese (ob/ob) mice have been described in thousands of publications. But the large prevalence of such internal islet lesions have never before been identified and visualised," says Ulf Ahlgren.
The researchers now want to study if similar intra-islet lesions also form in other models of type 2 diabetes and in humans, and if these may contribute to the diabetic phenotype.
Explore further: Lesions found within pancreatic islets provide clue for diabetes research
More information: Scientific Data, dataset: Spatial and quantitative datasets of the pancreatic -cell mass distribution in lean and obese mice. Authors: Saba Parween, Maria Eriksson, Christoffer Nord, Elena Kostromina and Ulf Ahlgren. DOI: 10.1038/sdata.2017.31
Scientific Reports, article: Intra-islet lesions and lobular variations in -cell mass expansion in ob/ob mice revealed by 3D imaging of intact pancreas. Authors: Saba Parween, Elena Kostromina, Christoffer Nord, Maria Eriksson, Per Lindstrm and Ulf Ahlgren. DOI: 10.1038/srep34885
Dryad Digital Repository, datasets from: Spatial and quantitative datasets of the pancreatic -cell mass distribution in lean and obese mice. Authors: Parween S, Eriksson M, Nord C, Kostromina E, Ahlgren U. DOI: 10.5061/dryad.pk8dv
Journal reference: Scientific Reports
Provided by: Umea University
Researchers at the Ume Center for Molecular Medicine have created the first 3D spatial visualization of an obese mouse pancreas showing the distribution dynamics of insulin producing beta cells. The results show significant ...
Pulses of the sugar glucose can restore normal insulin release in mouse pancreas cells that have been exposed to excess glucose, according to a study published in PLOS Computational Biology. This finding could improve understanding ...
Professor Ulf Ahlgren and associates at Umea University in Sweden are a leading research team in the world in the development of optical projection tomography. With the aid of this imaging technology, they have now described ...
(Medical Xpress) -- Research from Karolinska Institutet shows that insulin secretion in the pancreas is not under direct neural control, as has previously been thought. The few nerves that are present are connected to blood ...
Diabetes researchers at Sweden's Karolinska Institutet have developed a novel technique that makes it possible to monitor insulin resistance in a non-invasive manner over time in mice. The new method, presented in the journal ...
Researchers at Karolinska Institutet in Sweden have found an innovative way to study glucose regulation in the body: by transferring the vital insulin-producing cells from the pancreas to the eye, the latter can serve as ...
Please sign in to add a comment. Registration is free, and takes less than a minute. Read more
Link:
3-D visualization of the pancreasnew tool in diabetes research - Medical Xpress
Posted in Diabetes
Comments Off on 3-D visualization of the pancreasnew tool in diabetes research – Medical Xpress
Going gluten-free might actually increase your risk of diabetes – Men’s Fitness
Posted: March 14, 2017 at 2:46 pm
Bel Marra Health | Going gluten-free might actually increase your risk of diabetes Men's Fitness But those people who mistakenly think that going gluten-free is healthier may end up with higher risk of developing type-2 diabetes, say the researchers. Their study, which analyzed almost 200,000 people from three long-term studies and 4.24 million ... Type 2 diabetes linked to low gluten diets Could Going Gluten Free Raise Your Risk Of Diabetes? |
See the original post here:
Going gluten-free might actually increase your risk of diabetes - Men's Fitness
Posted in Diabetes
Comments Off on Going gluten-free might actually increase your risk of diabetes – Men’s Fitness
NAS Issues Report on Preparing for Future Products of Biotechnology: National Academies of Sciences, Engineering … – The National Law Review
Posted: March 13, 2017 at 8:44 pm
On March 9, 2017, the National Academies of Sciences, Engineering, and Medicine (NAS) announced the release (pre-publication version) of a new report:Preparing for Future Products of Biotechnology.Pursuant to the White House Office of Science and Technology Policy's (OSTP) July 2, 2015, memorandum, Modernizing the Regulatory System for Biotechnology Products, NAS was tasked with looking into the future and describing the possible future products of biotechnology that will arise over the next five to ten years, as well as providing some insights that can help shape the capabilities within the agencies as they move forward.
Via an ad hoc committee, the Committee on Future Biotechnology Products and Opportunities to Enhance Capabilities of the Biotechnology Regulatory System, NAS developed this report through several months of gathering and synthesizing information from several sources, including: 74 speakers over the course of three in-person meetings and eight webinars, including one presented by Lynn L. Bergeson; responses to its request for information from a dozen federal agencies; statements solicited from members of the public at its in-person meetings; written comments through the duration of the study; and recent NAS studies related to future products of biotechnology.
The report presents conclusions concerning the future biotechnology products themselves, as well the challenges that federal agencies will face in regulating them, which include:
The bioeconomy is growing rapidly and the U.S. regulatory system needs to provide a balanced approach for consideration of the many competing interests in the face of this expansion;
The profusion of biotechnology products over the next five to ten years has the potential to overwhelm the U.S. regulatory system, which may be exacerbated by a disconnect between research in regulatory science and expected uses of future biotechnology products;
Regulators will face difficult challenges as they grapple with a broad array of new types of bio-technology products -- for example, cosmetics, toys, pets, and office supplies -- that go beyond contained industrial uses and traditional environmental release;
The safe use of new biotechnology products requires rigorous, predictable, and transparent risk-analysis processes whose comprehensiveness, depth, and throughput mirror the scope, scale, complexity, and tempo of future biotechnology applications.
The report provides three recommendations for federal agencies in responding to these challenges, which it states should be taken to enhance the ability of the biotechnology regulatory system to oversee the consumer safety and environmental protection required for future biotechnology products:
The U.S. Environmental Protection Agency (EPA), the U.S. Food and Drug Administration (FDA), the U.S. Department of Agriculture (USDA), and other agencies involved in regulation of future bio-technology products should increase scientific capabilities, tools, expertise, and horizon scanning in key areas of expected growth of biotechnology, including natural, regulatory, and social sciences.
EPA, FDA, and USDA should increase their use of pilot projects to advance understanding and use of ecological risk assessments and benefit analyses for future biotechnology products that are unfamiliar and complex and to prototype new approaches for iterative risk analyses that incorporate external peer review and public participation.
The National Science Foundation, the Department of Defense, the Department of Energy, the National Institute of Standards and Technology, and other agencies that fund bio-technology research with the potential to lead to new biotechnology products should increase their investments in regulatory science and link research and education activities to regulatory-science activities.
The report is well-written and contains an impressive amount of new, relevant, and important information. The Committee participants are to be commended for an important new piece of scholarship in this area.
The reports conclusions are also significant, but not entirely unexpected.For those of us working in this space, we have recognized for years the lack of clarity regarding jurisdictional boundaries, the paucity of government resources, and the urgent need for regulatory clarity and significantly enhanced funding. Unfortunately, given current Trump Administration efforts to diminish government funding for EPA, FDA, and elsewhere, the well-crafted and spot-on recommendations may tragically fall on deaf ears.Shareholders should carefully review the report and work hard to ensure the recommendations are implemented. The consequences of failing to increase scientific capabilities, tools, expertise, and horizon scanning in key areas of expected growth of biotechnology, including natural regulatory, and social sciences -- the number one recommendation in the report -- are too great to ignore.
2017 Bergeson & Campbell, P.C.
See original here:
NAS Issues Report on Preparing for Future Products of Biotechnology: National Academies of Sciences, Engineering ... - The National Law Review
Posted in Biotechnology
Comments Off on NAS Issues Report on Preparing for Future Products of Biotechnology: National Academies of Sciences, Engineering … – The National Law Review
Puma Biotechnology Inc (PBYI) Moves Higher on Volume Spike for March 13 – Equities.com
Posted: March 13, 2017 at 8:44 pm
Market Summary Follow
Puma Biotechnology Inc is a A biopharmaceutical company
PBYI - Market Data & News
PBYI - Stock Valuation Report
Puma Biotechnology Inc (PBYI) traded on unusually high volume on Mar. 13, as the stock gained 11.86% to close at $44.80. On the day, Puma Biotechnology Inc saw 1.88 million shares trade hands on 13,031 trades. Considering that the stock averages only a daily volume of 1.15 million shares a day over the last month, this represents a pretty significant bump in volume over the norm.
Generally speaking, when a stock experiences a sudden spike in trading volume, it may be seen as a bullish signal for investors. An increase in volume means more market awareness for the company, potentially setting up a more meaningful move in stock price. The added volume also provides a level of support and stability for price advances.
The stock has traded between $73.27 and $19.74 over the last 52-weeks, its 50-day SMA is now $34.81, and its 200-day SMA $42.33. Puma Biotechnology Inc has a P/B ratio of 7.89.
Puma Biotechnology Inc is a biopharmaceutical company. It is engaged in the acquisition, development and commercialization of products to enhance cancer care.
Headquartered in Los Angeles, CA, Puma Biotechnology Inc has 160 employees and is currently under the leadership of CEO Alan H. Auerbach.
For a complete fundamental analysis analysis of Puma Biotechnology Inc, check out Equities.coms Stock Valuation Analysis report for PBYI.
Want to invest with the experts? Subscribe to Equities Premium newsletters today! Visit http://www.equitiespremium.com/ to learn more about Guild Investments Market Commentary and Adam Sarhans Find Leading Stocks today.
To get more information on Puma Biotechnology Inc and to follow the companys latest updates, you can visit the companys profile page here: PBYIs Profile. For more news on the financial markets and emerging growth companies, be sure to visit Equities.coms Newsdesk. Also, dont forget to sign-up for our daily email newsletter to ensure you dont miss out on any of our best stories.
All data provided by QuoteMedia and was accurate as of 4:30PM ET.
DISCLOSURE: The views and opinions expressed in this article are those of the authors, and do not represent the views of equities.com. Readers should not consider statements made by the author as formal recommendations and should consult their financial advisor before making any investment decisions. To read our full disclosure, please go to: http://www.equities.com/disclaimer
The rest is here:
Puma Biotechnology Inc (PBYI) Moves Higher on Volume Spike for March 13 - Equities.com
Posted in Biotechnology
Comments Off on Puma Biotechnology Inc (PBYI) Moves Higher on Volume Spike for March 13 – Equities.com
Portugal to support Nigeria in Biotechnology activities – TV360 Nigeria – TV360
Posted: March 13, 2017 at 8:44 pm
The Portuguese Minister of Science and Technology, Manuel Heitor, say his country will offer technical support to Nigeria in key biotechnology activities that will enhance tropical agriculture and food security.
Heitor made this known in a statement made available to the News Agency of Nigeria (NAN) by the Head, Protocol and Communications, National Biotechnology Development Agency (NABDA), Ifeoma Ndefo on Monday in Abuja.
The minister visited the Director-General, National Biotechnology Development Agency (NABDA), LucyOgbadu, with a team of experts in different fields of biotechnology from various institutions in Portugal.
In his address, Heitor indicated interest of the Portuguese delegation in partnering with the NABDA on efforts to expand technologies that would enhance tropical agriculture and food security.
The minister,who is also the minister of Education, declared the interest of his country in assisting NABDA scientific officers wishing to acquire Masters and P.HD degrees in Portugal.
He identified areas of NABDAs activities akin to their fields of competencessuch as cancer prevention, stem cell research, Biogas production (converting wastes to generate electricity) and bio-remediation.
Heitor said all of thesewere areas of interest for collaboration as well as knowledge sharing between their institutions and NABDA.
On her part, Director-General, NABDA, commended the minister and his team for their interest to partner with the Agency and to ensure that Nigeria reaps the potential benefit that biotechnology offers.
The NABDA boss highlighted the various projects and activities of the five technical departments of the Agency.
This included the six Centres of Excellence located at frontline universities of the each of the Six Geo-political Zones of the nation (Uni-Jos, ABU Zaria, Uni- Port, UNN, Uni-Ibadan and Uni- Maiduguri).
Others are the NABDAs Bioresources Centres (BioDecs) spread across twenty-five states of the federation.
The News Agency of Nigeria recalls that the agency was established by the Federal Government to implement policy aimed at promoting, coordinating, and setting research and development priority in biotechnology for Nigeria.
NAN.
The rest is here:
Portugal to support Nigeria in Biotechnology activities - TV360 Nigeria - TV360
Posted in Biotechnology
Comments Off on Portugal to support Nigeria in Biotechnology activities – TV360 Nigeria – TV360
Woodrose Ventures Corporation Announces Proposed Acquisition of Global Stem-Cell Biotechnology Company … – Marketwired (press release)
Posted: March 13, 2017 at 8:43 pm
VANCOUVER, BRITISH COLUMBIA--(Marketwired - March 13, 2017) -
NOT FOR DISSEMINATION IN THE UNITED STATES
Editors Note: There is a photo associated with this press release.
Woodrose Ventures Corporation (TSX VENTURE:WRS.H) ("Woodrose" or the "Company") is pleased to announce that it has entered into an agreement (the "Agreement") dated March 10, 2017 to acquire all of the shares of Novoheart Holdings Ltd. ("Novoheart"), a global stem cell biotechnology company dedicated to human heart engineering (the "Transaction"). Novoheart develops products and provides services focused on engineering prototypes of bio-artificial human heart tissues and chambers for drug discovery, cardiotoxicity screening, disease modelling and therapeutic applications.
The Transaction will constitute a "reverse-takeover" of Woodrose in accordance with the policies of the TSX Venture Exchange (the "TSXV") and the reactivation of Woodrose, which is currently a NEX-listed issuer.
About Novoheart
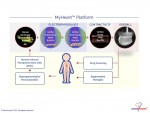
Novoheart is a global stem cell biotechnology company headquartered in Hong Kong with R&D Innovation Centres being set up in the United States. Novoheart's mission is to revolutionize drug discovery and the development of heart therapeutics with its range of proprietary bioengineered human heart constructs, collectively known as the MyHeart platform, and to further develop them into transplantable heart grafts for cell-based regenerative therapies with superior safety and efficacy. Its scientific team has pioneered a range of best-in-class bioengineering technologies and constructed the world's first human mini-heart "novoHeart" with which the Novoheart team intends to revolutionize:
1) Pre-clinical drug discovery, cardiotoxicity screening and heart disease modelling;
2) Post-discovery, clinical development of novel therapeutics; and
3) Pre-clinical and clinical development of cell-based cardiac regenerative therapies.
Novoheart's immediate focus is to innovate and accelerate the lengthy, expensive and inefficient drug development process. The development of a new drug candidate typically costs US$2-4bn and takes 10+ years (Tufts Centre for the Study of Drug Development, Tufts CSDD R&D Cost Study 2014) with extremely poor success rates of
Novoheart's intellectual property portfolio, including the human "heart-in-a-jar" (novoHeart) and other related next-generation technologies of the MyHeart platform (see figure below) are unique solutions that help bridge the gap between pre-clinical and clinical drug trials. The MyHeart platform provides advanced human heart surrogates for pre-screening of drug formulas and the elimination of toxic compounds early on in the drug development process, minimizing the risk towards patients. Significantly, the MyHeart Platform provides real time data on the effects of drug formulations enabling drug development companies to undertake "on-the-fly" reformulation of drug candidates to optimize efficacy and toxicological profiles. With Novoheart's technologies, we aim to significantly reduce pre-clinical R&D time and costs, and importantly, improve trial successes. It is anticipated that drug screening results using Novoheart's human engineered tissues would be accepted as reliable indicators for toxicity and efficacy, thereby qualifying the test compounds for accelerated drug development.
Novoheart adopts a hybrid business model by:
These products and services are designed to significantly reduce the time, cost, and use of animal models, as well as improve patient safety, and facilitate pharmaceutical discovery and development. Novoheart is currently working with leading academic and pharmaceutical partners to innovate drug discovery and toxicity screening protocols. Our targeted clients are pharmaceutical companies, government units, and research institutions.
Novoheart was incorporated in 2014 pursuant to the laws of British Virgin Islands (BVI) and its controlling shareholder is Medera Group Limited, a BVI entity. Novoheart has one wholly owned Hong Kong subsidiary "Novoheart Limited" ("Novoheart Hong Kong") which is the group operating entity.
Novoheart Hong Kong was incorporated in January 2014 by founder and CEO Prof. Ronald Li, with scientific co-founders Prof. Kevin Costa and Prof. Michelle Khine.
Novoheart's foundational technologies are the direct outcome of over 15 years of research effort supported by R&D investments amounting to approximately USD30MM. These research efforts, performed at Johns Hopkins University, Icahn School of Medicine at Mount Sinai, University of California Irvine, University of California Davis, and the University of Hong Kong by our scientific founders, have received major recognitions such as American Heart Association's Best Study of 2005, Ground-breaking Study of 2006, and Late-breaking Studies of 2002, 2003, 2005 and 2007, and the Spirit of Hong Kong Innovating for Good Award in 2015. The "human-heart-in-a-jar" technology was selected by Google's Solve For X as a Moonshot Project in 2015.
Novoheart's scientific founders and advisors are renowned pioneering leaders in the stem cell and cardiac space, with a successful track record in developing and commercializing ground-breaking technologies. In September 2014, Novoheart established its R&D base and office in the Hong Kong Science Park, where it continues to innovate solutions for drug discovery and human heart tissue engineering.
In December 2014, Novoheart signed a strategic partnership with a major global pharmaceutical company (the "Global Pharma Partner") headquartered in New York City to validate the MyHeart platform. The success of this validation process has resulted in follow on income-generating projects.
In January 2015, Novoheart's R&D proposal to develop bio-artificial heart tissues for drug screening received 50/50 matched funding from the Innovation & Technology Commission (ITC) of the Government of Hong Kong, with a total project cost of over HK$21MM over 2 years. It was also the largest biotech project granted by ITC for that year. Novoheart owns all of the intellectual property generated from this project, and as a result of the R&D, Novoheart has applied or is in the application process for 3 new patents covering newly developed technology, including the human ventricular cardiac anisotropic sheet (hvCAS) as a powerful tool for detecting drug-induced arrhythmias with the results published in the prestigious international peer-reviewed bioengineering journal Advanced Materials (Shum et al. 2017, Advanced Materials, 29). Additionally, Novoheart holds exclusive worldwide licenses or options to acquire the same for technologies that constitute its MyHeart platform and future developments.
In December 2015, Novoheart signed a second contract with the Global Pharma Partner to build disease-specific engineered human heart tissues and chambers for drug discovery. The total project cost is US$726,000 over 1.5 years.
In February 2017, the Corporate Venture Fund (CVF) of the Hong Kong Science and Technology Parks Corporation (HKSTPC) completed an equity investment of approximately US$250,000 into Novoheart and an additional investment would be made at the Transaction.
Novoheart Financial Information
The following table includes a summary of certain financial information of Novoheart and is derived from its financial statements for the years ended June 30, 2016 and June 30, 2015.
Summary of the Transaction
Under the terms of the Agreement, the shareholders of Novoheart will receive an aggregate of 66,086,600 common shares of Woodrose on a post-Consolidation basis (see below) ("Woodrose Post-Consolidation Shares"). In addition, a finder's fee of 2,313,038 Woodrose Post-Consolidation Shares will be paid to Cynosure Private Equity Limited in connection with the Transaction.
In connection with the Transaction, Woodrose intends to complete a consolidation of all its outstanding common shares on the basis of 3.56878449 old common shares for each one new common share (the "Consolidation"). In addition, Woodrose intends to complete a non-brokered private placement (the "Private Placement") of 11,700,000 subscription receipts ("Subscription Receipts") at a price of CDN$0.50 per Subscription Receipt to raise gross proceeds of CDN$5,850,000, which will be held in escrow in accordance with the terms of a subscription receipt agreement (the "Subscription Receipt Agreement"). It is anticipated that the Subscription Receipt Agreement will provide that, upon completion of the Transaction, each Subscription Receipt will automatically convert into one Woodrose Post-Consolidation Share. The Subscription Receipt Agreement will also provide that, in the event the Transaction is terminated or does not complete within an agreed timeframe, the Subscription Receipts will be cancelled and the funds will be returned to the holders. Woodrose may pay cash fees in an amount not to exceed 7% of the gross proceeds (to a maximum of $364,000) to certain finders involved in the Private Placement and may issue finder's warrants ("Finder's Warrants"), in an amount not to exceed 7% of the number of Subscription Receipts issued (to a maximum of 728,000 Finders Warrants) each of which would entitle the holder to acquire one Woodrose Post-Consolidation Share at a price of CDN$0.50 for a period of two years following closing of the Private Placement. All securities issued pursuant to the Private Placement will be subject to a statutory hold period of four months and one day.
The Company intends to use the net proceeds of the offering to finance investment in drug discovery and screening, establish commercial partnerships, expand the current laboratory, hire additional research and development team members and for working capital and general corporate purposes.
Upon completion of the Transaction, it is anticipated that the Company will be classified as a Tier 2 Technology Issuer on the TSXV and will change its name to "Novoheart Holdings (BC) Limited" or such other name as is acceptable to the Board of Directors. Closing of the Transaction ("Closing") is subject to conditions precedent, that include, but are not limited to, the following:
The Transaction is an "arm's length" transaction (as defined by the policies of the TSXV). Woodrose intends to rely an exemption from the sponsorship requirements of the policies of the TSXV.
Proposed Management Team
Upon closing of the Transaction, the following directors and senior officers are anticipated to be appointed in replacement of Woodrose's current board and management:
Prof. Ronald Li, B.Sc. (Hons), Ph.D. (Proposed President, Chief Executive Officer and Director)
Prof. Ronald Li is a co-founder of Novoheart, and has been serving as the CEO since 2016. He is concurrently Director of Ming-Wai Lau Centre for Reparative Medicine, HK node, Karolinska Institutet (KI), Sweden, with a professorial cross appointment at the Dr. Li Dak-Sum Research Centre, The University of Hong Kong (HKU)-KI Collaboration in Regenerative Medicine of HKU. Prof. Li has been an advocate of stem cell technology for many years, starting from his career as Assistant Professor of Cardiology, and Cellular and Molecular Medicine at the Johns Hopkins University (JHU) School of Medicine. He founded and led the Human Embryonic Stem Cell Consortium when he was recruited in 2005 to become a tenured Associate Professor at the University of California, Davis, in light of state's USD3-billion stem cell initiative Proposition 71. Prof. Li was the Founding Director of the Stem Cell & Regenerative Medicine Consortium (SCRMC) at the University of Hong Kong (HKU) from 2010 to 2015. He also co-directed the Section of Cardiovascular Cell & Tissue Engineering in Icahn School of Medicine at Mount Sinai with Prof. Kevin Costa. Prof. Li has received multiple accolades and recognitions during his career, including the Spirit of Hong Kong Innovating for Good Award by the South China Morning Post (2015), the Top Young Faculty Award (2002, 2004), the Top Prize for the Young Investigator Basic Research (2001) and Top Postdoctoral Fellow Helen Taussig Award (2001) of JHU School of Medicine, Young Investigator Award 1st Prize from the Heart Rhythm Society (2002), and the Career Development Award from the Cardiac Arrhythmias Research & Education Foundation (2001).
Prof. Li graduated with his B.S. with honors in Biotechnology from University of Waterloo, Ontario, on Dean's List and his Ph.D. in Cardiology/Physiology at the University of Toronto.
Dr. Camie Chan, B.Sc. (Hons), M.Sc., Ph.D. (Proposed Chief Operating Officer and Director)
Dr. Camie Chan joined Novoheart Hong Kong as the Chief Operating Officer in 2016, after having served at HKU as the Deputy Director of the Faculty of Medicine Core Facility, a founding member of the Management Committee of the Stem Cell & Regenerative Medicine Consortium (SCRMC), and Assistant Professor in the Department of Anatomy, between 2010 and 2016. She has had extensive experience managing laboratory operations in her capacity at HKU, and her prior career as Assistant Professor at the University of California, Davis, and Assistant Investigator at the Shriners Hospital for Children. Dr. Chan is also a co-inventor of technology allowing mass production of human ventricular heart cells from pluripotent stem cells.
Dr. Chan graduated with her B.Sc. with honors at the University of Waterloo, followed by obtaining her M.Sc. degree in Medical Sciences and Ph.D. degree in Immunology at the University of Toronto, Canada. She then received postdoctoral training at the Sydney Kimmel Cancer Research Center at the Johns Hopkins University. She has garnered numerous awards in her career, including the prestigious National Institute of Allergy and Infectious Diseases (NIAID) Developmental Research Grant Award.
Prof. Kevin Costa, B.S., Ph.D. (Proposed Chief Scientific Officer)
Prof. Costa is Director of the Section of Cardiovascular Cell and Tissue Engineering at the Icahn School of Medicine at Mount Sinai in New York City. Prof. Costa was previously trained at the Johns Hopkins University and on the faculty as Associate Professor of Biomedical Engineering at Columbia University. As a "blue-blood" biomedical engineering (BME) expert (B.S. and M.S. in BME from Boston University, Ph.D. in BME from UC San Diego, and postdoc in BME from JHU and University of Washington) in cell and tissue biomechanics and cardiac tissue engineering, he has developed one of the first engineered cardiac tissue systems. Since 2009, he has been working with Prof. Ronald Li to translate such systems into human cells. Prof. Costa has received research funding from the Whitaker Foundation, the National Science Foundation (NSF) and the National Institutes of Health (NIH; NHLBI, NIBIB, and NIGMS). He was also a recipient of the prestigious Faculty Early Career Development (CAREER) Award from the NSF. Prof. Costa is an inventor of several cardiac tissue engineering technologies and one of the scientific co-founders of Novoheart Hong Kong.
Ms. Iris Lo, B. Comm. (Hons), CPA, CA (Proposed Chief Financial Officer)
Ms. Lo is a seasoned professional with expertise in corporate finance, mergers and acquisitions, accounting, and finance. Prior to joining Novoheart, Ms. Lo was the Director of Corporate Development & Analysis at Cardiome Pharma Corp., a Canadian public company dually listed on the TSX and NASDAQ (TSX: COM, NASDAQ: CRME). At Cardiome, she held responsibilities in equity and debt financing, corporate mergers and acquisitions, product licensing and distributions, financial planning and analysis, as well as regulatory and risk management. During her tenure at Cardiome, Ms. Lo participated in transactions totaling over US$240 million as Cardiome grew from a company with a market capitalization of US$25 million to over US$150 million at its peak. She brings with her valuable experience from the life sciences and pharmaceutical sector, as well as expertise in dealing with the complexities of operating and financing public corporations. Ms. Lo was also previously a Manager in the Transaction Services team at PwC Hong Kong and began her career articling with KPMG Vancouver. She is a Chartered Professional Accountant and holds a Bachelor of Commerce (Honours) from the Sauder School of Business at the University of British Columbia.
Mr. Victor Chang (Proposed Director)
Mr. Chang is a seasoned investor who has lately become focused on start-ups. Mr. Chang started his career with Lippo Securities Limited in 1996 and became a Director of Grand International Holdings Limited in 1999, which was engaged in general investments. During the period from 2007 to 2009, he was a Director and Responsible Officer for Astrum Capital Management Limited carrying out regulated activities under the Securities and Futures Ordinance ("SFO", Cap. 571, Laws of Hong Kong) and with Murtsa Capital Partners Limited as well. During the period from 2007 to 2012, he was also a compliance consultant for Astrum Capital Management Limited. As co-founder and Managing Director of Zebra Strategic Outsource Solution, he has over 16 years of experience in recruitment process outsourcing, executive search as well as and private investment management. In Apil 2013, he successfully brought Zebra Strategic Holdings Limited which offers holistic HR solutions to IPO on the HK GEM board (Stock Code: 8260) and was re-designated as and is currently a Non-Executive Director with the company. He is currently a Director and Responsible Officer of Dakin Financial Group, a corporation licensed to carry out type 1, 2 & 9 regulated activity under the Hong Kong Securities and Futures Ordinance.
Mr. Tong Ricky Chiu (Proposed Director)
As a key founder and visionary for Grand Power Logistics Group Inc., which was listed on the TSX Venture Exchange (GPW.V) before its privatization in 2016, and Baoshinn International Express Ltd., Mr. Chiu adds value with his immense corporate development and growth skills. He received his education in Oxford University, England, and Beijing University, and began his career in Australia. He has a diversified background in a wide range of industries with roles in finance, audit, real estate, merchandise trading and travel, as well as logistics.
Mr. James Topham (Proposed Director)
Mr. Topham is an experienced executive with expertise in finance, accounting, auditing and entrepreneurial technology companies. He was an audit partner leading KPMG's Technology Group in the Vancouver office for 20 years where he worked with many fast growing public companies and was involved in many M&A and IPO transactions in Canada, the US and Europe. Mr. Topham founded Social Venture Partners Vancouver in 2001 with a mission to strengthen the organizational capacity of innovative non-profits serving children in-need and youth at-risk. It has funded several million dollars and provided thousands of hours of executive time mentoring these local non-profits. Since retiring at KPMG 7 years ago, Mr. Topham has worked on several Boards of both public and private technology companies. He received a lifetime achievement award from the BC Technology Industry Association and was awarded the designation of Fellow Chartered Public Accountant (FCPA) from the Chartered Public Accountants of BC for his career achievements in the profession and community. He was a founder and Board member for 9 years of the BC Technology Industry Association that represents the technology industry in BC. Mr. Topham is a CPA and has a Bachelor of Commerce degree with Honours from the University of Saskatchewan graduating as the most distinguished graduate in the College of Commerce.
Mr. Allen Ma (Proposed Director)
As a 30-year technology industry veteran, Mr. Ma was the CEO of Hong Kong Science & Technology Parks before he retired in July 2016. He held senior executive positions within the information and communications technology sector. His past roles include president for Asia-Pacific at British Telecom, vice-president for Asia at the global telecom solutions sector of Motorola, executive director of Hong Kong Telecommunications - subsequently called Cable & Wireless HKT - and managing director of Hong Kong Telecom CSL. Ma holds an MBA from the University of Toronto and is a fellow member of both the Chartered Institute of Management Accountants, UK and the Association of Chartered Certified Accountants, UK. He is also a Certified Management Accountant of Canada.
Proposed Advisory Team
Novoheart is supported by a Scientific Advisory Board whose proposed composition consists of eminent scientists renowned in the fields of stem cells, cardiac biology and physiology, tissue engineering, and clinical cardiology including clinical trials research, from top academic research institutes in the U.S.A. Their technical expertise will guide the development of Novoheart as a forerunner in the application of cutting-edge technologies to develop new and better treatments for heart disease and beyond.
Further Details
Both the Company and Novoheart intend to work diligently to complete the conditions precedent to Closing and anticipate completion of the Transaction in the second quarter of 2017. The Company will update its shareholders with further details as they become available.
ON BEHALF OF WOODROSE VENTURES CORPORATION
Darren Devine, President, CEO and Director
NEITHER THE TSX VENTURE EXCHANGE NOR ITS REGULATION SERVICES PROVIDER (AS THAT TERM IS DEFINED IN THE POLICIES OF THE TSX VENTURE EXCHANGE) ACCEPTS RESPONSIBILITY FOR THE ADEQUACY OR ACCURACY OF THIS RELEASE.
Completion of the Transaction is subject to a number of conditions, including but not limited to, Exchange acceptance and if applicable pursuant to Exchange requirements, majority shareholder approval. Where applicable, the Transaction cannot close until the required shareholder approval is obtained. There can be no assurance that the Transaction will be completed as proposed or at all.
Investors are cautioned that, except as disclosed in the Filing Statement to be prepared in connection with the Transaction, any information with respect to the Transaction may not be accurate or complete and should not be relied on. Trading in securities of the Company should be considered highly speculative.
The TSX Venture Exchange has in no way passed upon the merits of the Transaction and has neither approved nor disproved the contents of this news release.
Cautionary Note Regarding Forward-Looking Statements
Information set forth in this news release may involve forward-looking statements under applicable securities laws. Forward-looking statements are statements that relate to future, not past, events. In this context, forward-looking statements often address expected future business and financial performance, and often contain words such as "anticipate", "believe", "plan", "estimate", "expect", and "intend", statements that an action or event "may", "might", "could", "should", or "will" be taken or occur, or other similar expressions. All statements, other than statements of historical fact, included herein including, without limitation; statements about the terms and completion of the Transaction are forward-looking statements. By their nature, forward-looking statements involve known and unknown risks, uncertainties and other factors which may cause the actual results, performance or achievements, or other future events, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Such factors include, among others, the following risks: failure to satisfy all conditions precedent to the Transaction, including shareholder approval, approval of the TSX Venture Exchange and completion of the necessary financings and the additional risks identified in the management discussion and analysis section of Woodrose Corporation's interim and most recent annual financial statement or other reports and filings with the TSX Venture Exchange and applicable Canadian securities regulators. Forward-looking statements are made based on management's beliefs, estimates and opinions on the date that statements are made and the respective companies undertakes no obligation to update forward-looking statements if these beliefs, estimates and opinions or other circumstances should change, except as required by applicable securities laws. Investors are cautioned against attributing undue certainty to forward-looking statements.
To view the photo associated with this press release, please visit the following link: http://www.marketwire.com/library/20170312-1088577_MyHeart_800.jpg
Originally posted here:
Woodrose Ventures Corporation Announces Proposed Acquisition of Global Stem-Cell Biotechnology Company ... - Marketwired (press release)
Posted in Biotechnology
Comments Off on Woodrose Ventures Corporation Announces Proposed Acquisition of Global Stem-Cell Biotechnology Company … – Marketwired (press release)
Vitamin C can target and kill cancer stem cells, study shows … – Medical News Today
Posted: March 13, 2017 at 8:43 pm
Cancer is currently one of the top killers worldwide, and the number of cancer cases is only expected to rise. Although there are a number of therapies available, most of them are toxic and cause serious side effects. New research examines the impact of the natural vitamin C on cancer cell growth.
Cancer is the second leading cause of death and disease worldwide, accounting for almost 9 million deaths in 2015, according to the World Health Organization (WHO).
The global number of new cases of cancer are expected to grow by around 70 percent in the next 20 years.
In the United States, the National Cancer Institute (NCI) estimate that almost 40 percent of U.S. men and women will have developed cancer at one point during their lives.
There are various treatment options available for cancer, but they are not always effective; most of them are toxic, and they tend to have a variety of side effects.
In some more aggressive cases, the cancer does not respond to treatment, and it is believed that cancer stem-like cells are the reason why the cancer comes back and metastasizes.
New research, published in the journal Oncotarget, examines the effectiveness of three natural substances, three experimental drugs, and one clinical drug in stopping the growth of these cancer stem cells (CSCs.)
The study was conducted by researchers from the University of Salford in Manchester in the United Kingdom, and was led by Dr. Gloria Bonuccelli.
In total, the researchers measured the impact of seven substances: the clinical drug stiripentol, three experimental drugs (actinonin, FK866, and 2-DG), and three natural substances (caffeic acid phenyl ester (CAPE), silibinin, and ascorbic acid (vitamin C).)
The research focused on the bioenergetic processes of CSCs, which enable the cells to live and multiply. The study aimed to disrupt the CSCs' metabolism and ultimately prevent their growth.
Of all the substances tested, the team found that actinonin and FK866 were the most effective. However, the natural products were also found to prevent the formation of CSCs, and vitamin C was 10 times more effective than the experimental drug 2-DG.
Additionally, the study revealed that ascorbic acid works by inhibiting glycolysis - the process by which glucose is broken down within the cell's mitochondria and turned into energy for the cell's proliferation.
Dr. Michael P. Lisanti, professor of translational medicine at the University of Salford, comments on the findings:
"We have been looking at how to target cancer stem cells with a range of natural substances including silibinin (milk thistle) and CAPE, a honey-bee derivative, but by far the most exciting are the results with vitamin C. Vitamin C is cheap, natural, nontoxic and readily available so to have it as a potential weapon in the fight against cancer would be a significant step."
"This is further evidence that vitamin C and other nontoxic compounds may have a role to play in the fight against cancer," says the study's lead author.
"Our results indicate it is a promising agent for clinical trials, and as an add-on to more conventional therapies, to prevent tumor recurrence, further disease progression, and metastasis," Bonuccelli adds.
Vitamin C has been shown to be a potent, nontoxic, anticancer agent by Nobel Prize winner Linus Pauling. However, to the authors' knowledge, this is the first study providing evidence that ascorbic acid can specifically target and neutralize CSCs.
Learn how 300 oranges' worth of vitamin C can impair cancer cells.
Read more here:
Vitamin C can target and kill cancer stem cells, study shows ... - Medical News Today
Posted in Cell Medicine
Comments Off on Vitamin C can target and kill cancer stem cells, study shows … – Medical News Today
